
The optical image of TN J0924-2201, a very distant radio galaxy at (redshift) z = 5.19, obtained with the Hubble Space Telescope. (c) NASA/STScI/NAOJ.
For us carbon-based life forms, carbon is a fairly important part of the chemical makeup of the Universe. However, carbon and oxygen were not created in the Big Bang, but rather much later in stars. How much later? In a surprising find, scientists have detected carbon much earlier in the Universe’s history than previously thought.
Researchers from Ehime University and Kyoto University have reported the detection of carbon emission lines in the most distant radio galaxy known. The research team used the Faint Object Camera and Spectrograph (FOCAS) on the Subaru Telescope to observe the radio galaxy TN J0924-2201. When the research team investigated the detected carbon line, they determined that significant amounts of carbon existed less than a billion years after the Big Bang.
How does this finding contribute to our understanding of the chemical evolution of the universe and the possibilities for life?
To understand the chemical evolution of our universe, we can start with the Big Bang. According to the Big Bang theory, our universe sprang into existence about 13.7 billion years ago. For the most part, only Hydrogen and Helium ( and a sprinkle of Lithium) existed.
So how do we end up with everything past the first three elements on the periodic table?
Simply put, we can thank previous generations of stars. Two methods of nucleosythesis (element creation) in the universe are via nuclear fusion inside stellar cores, and the supernovae that marked the end of many stars in our universe.
Over time, through the birth and death of several generations of stars, our universe became less “metal-poor” (Note: many astronomers refer to anything past Hydrogen and Helium as metals”). As previous generations of stars died out, they “enriched” other areas of space, allowing future star-forming regions to have conditions necessary to form non-star objects such as planets, asteroids, and comets. It is believed that by understanding how the universe created heavier elements, researchers will have a better understanding of how the universe evolved, as well as the sources of our carbon-based chemistry.
So how do astronomers study the chemical evolution of our universe?
By measuring the metallicity (abundance of elements past Hydrogen on the periodic table) of astronomical objects at various redshifts, researchers can essentially peer back into the history of our universe. When studied, redshifted galaxies show wavelengths that have been stretched (and reddened, hence the term redshift) due to the expansion of our universe. Galaxies with a higher redshift value (known as “z”) are more distant in time and space and provide researchers information about the metallicity of the early universe. Many early galaxies are studied in the radio portion of the electromagnetic spectrum, as well as infra-red and visual.
The research team from Kyoto University set out to study the metallicity of a radio galaxy at higher redshift than previous studies. In their previous studies, their findings suggested that the main era of increased metallicity occurred at higher redshifts, thus indicating the universe was “enriched” much earlier than previous believed. Based on the previous findings, the team then decided to focus their studies on galaxy TN J0924-2201 – the most distant radio galaxy known with a redshift of z = 5.19.
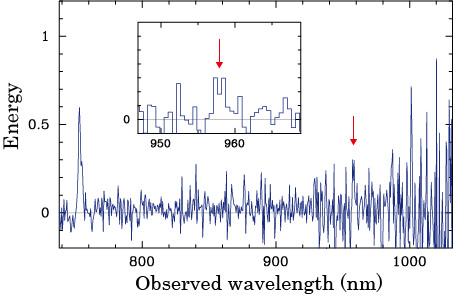
The deep optical spectrum of TN J0924-2201 obtained with FOCAS on the Subaru Telescope. The red arrows point to the carbon emission line.
The research team used the FOCAS instrument on the Subaru Telescope to obtain an optical spectrum of galaxy TN J0924-2201. While studying TN J0924-2201, the team detected, for the first time, a carbon emission line (See above). Based on the detection of the carbon emission line, the team discovered that TN J0924-2201 had already experienced significant chemical evolution at z > 5, thus an abundance of metals was already present in the ancient universe as far back as 12.5 billion years ago.
If you’d like to read the team’s findings you can access the paper Chemical properties in the most distant radio galaxy – Matsuoka, et al at: http://arxiv.org/abs/1107.5116
Source: Universe Today
No hay comentarios:
Publicar un comentario